Case Study
Move-MORE Study
The Move-MORE study is a pilot clinical research project conducted through the Helfgott Research Institute at National University of Natural Medicine (NUNM), designed to evaluate the feasibility, acceptability, and preliminary outcomes of a multicomponent mindfulness and movement intervention for individuals living with lumbosacral radiculopathy (sciatica), a chronic pain condition involving nerve-related leg pain.


Overview
The Move-MORE study sits at the intersection of integrative medicine, behavioral science, and digital health, with the overarching aim of developing a scalable, person-centered program for chronic pain management.
In parallel with the clinical research, strategic documentation and communication materials were developed to support participant understanding and research dissemination. Ensuring clarity and transparency throughout the research lifecycle, from informed consent to manuscript preparation, demanded careful attention to established clinical research reporting standards such as SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) and GRIPP-2 (Guidance for Reporting Involvement of Patients and the Public).
Client
Wexler Lab, Helfgott Research Institute
My Role
Clinical Research Assistant
Date
July 2025 – Present
Approach & Contributions
In collaboration with the Move-MORE research team, our role encompassed developing visual and written materials that both supported rigorous study conduct and enhanced participant and scientific audience engagement. This included:
-
Informed Consent Presentation & Video Materials: Translating complex study design, risks, and participant expectations into accessible language and visuals.
-
Workflow Visualizations: Mapping participant flows, data collection touchpoints, and study visit sequences into clear visual frameworks.
-
Conference Posters: Designing visually compelling posters for national and international scientific meetings to communicate study rationale, methods, and emerging results.
-
Manuscript Development: Structuring clinical manuscripts to articulate study protocol, results, and implications in alignment with SPIRIT for clinical trial protocols and GRIPP-2 for reporting any patient/public involvement components.

The Results
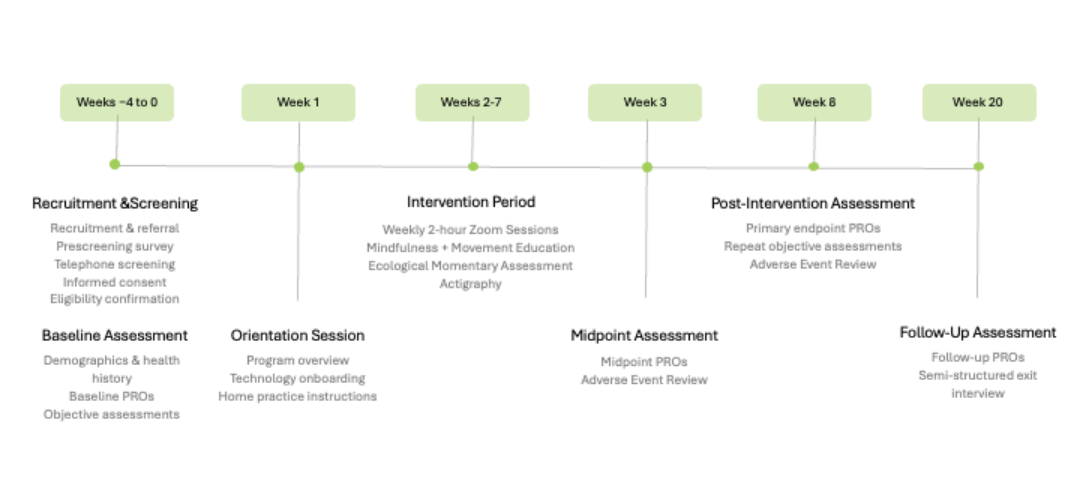
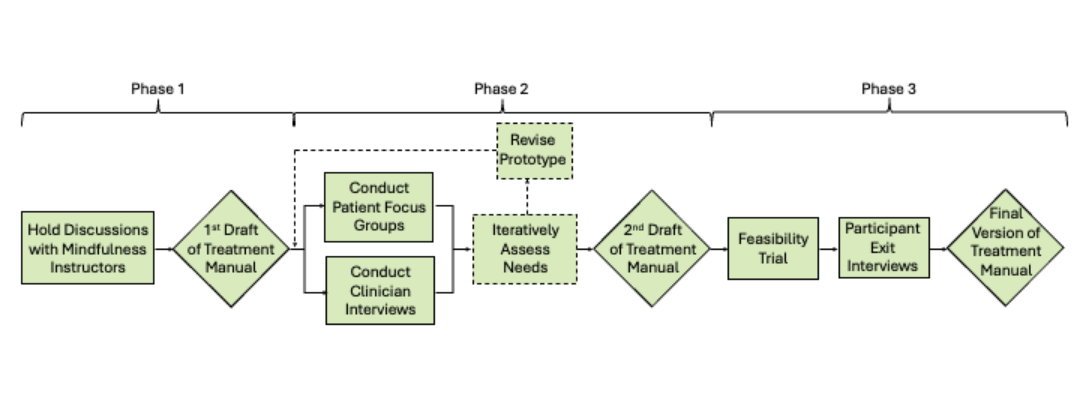
The study’s success hinged on clearly communicating what participation entailed: the intervention elements (mindfulness, movement, weekly sessions), data collection modalities (daily check-ins, wearable activity tracking), and participant responsibilities. To facilitate this, we storyboarded and scripted an informed consent video that paired plain-language narration with supportive visuals, breaking down the trial’s structure into digestible segments.
The video supplemented traditional text-based consent forms, offering prospective participants a consistent and trustworthy introduction to the study. Visual metaphors and motion graphics were used to illustrate the core components of the Move-MORE intervention and participant expectations, helping viewers make fully informed decisions about joining.
At major scientific meetings, presenting Move-MORE findings required posters that balanced research data with visual clarity.
We designed posters that foregrounded the study’s multicomponent design, articulating hypotheses, methods, and preliminary results in structured, reader-friendly sections.
The design and layout facilitated rapid comprehension for conference attendees, enabling lively discussion and feedback that enriched subsequent manuscript drafts.
Writing for peer-reviewed journals demanded strict adherence to clinical research reporting norms. Using the SPIRIT checklist as a guiding framework, we helped organize the protocol and results manuscripts to systematically address essential trial elements, from eligibility criteria and intervention descriptions to outcomes and statistical plans. Concurrently, attention to GRIPP-2 ensured transparent reporting on any patient or public involvement elements embedded in the research design.
On Til Morning.
Location
Portland, Oregon
